Following our recent webinar on access to cystic fibrosis modulators, we received a number of important questions from families and patients. We've compiled the answers here so that everyone can benefit from the information.
Firstly, we’ve had some questions about dosage of Triko and Bexdeco.
As many of you will know, triple-combination CF modulator treatments usually include two different tablets, taken in the morning and evening.
For adults and children aged 6-12 (who are over 30kg in weight) the dose is:
Two tablets taken in the morning (containing elexacaftor 100mg, tezacaftor 50mg and ivacaftor 75mg)
One tablet taken in the evening (containing ivacaftor 150mg only)
For the products produced by Beximco, these are sold separately, as:
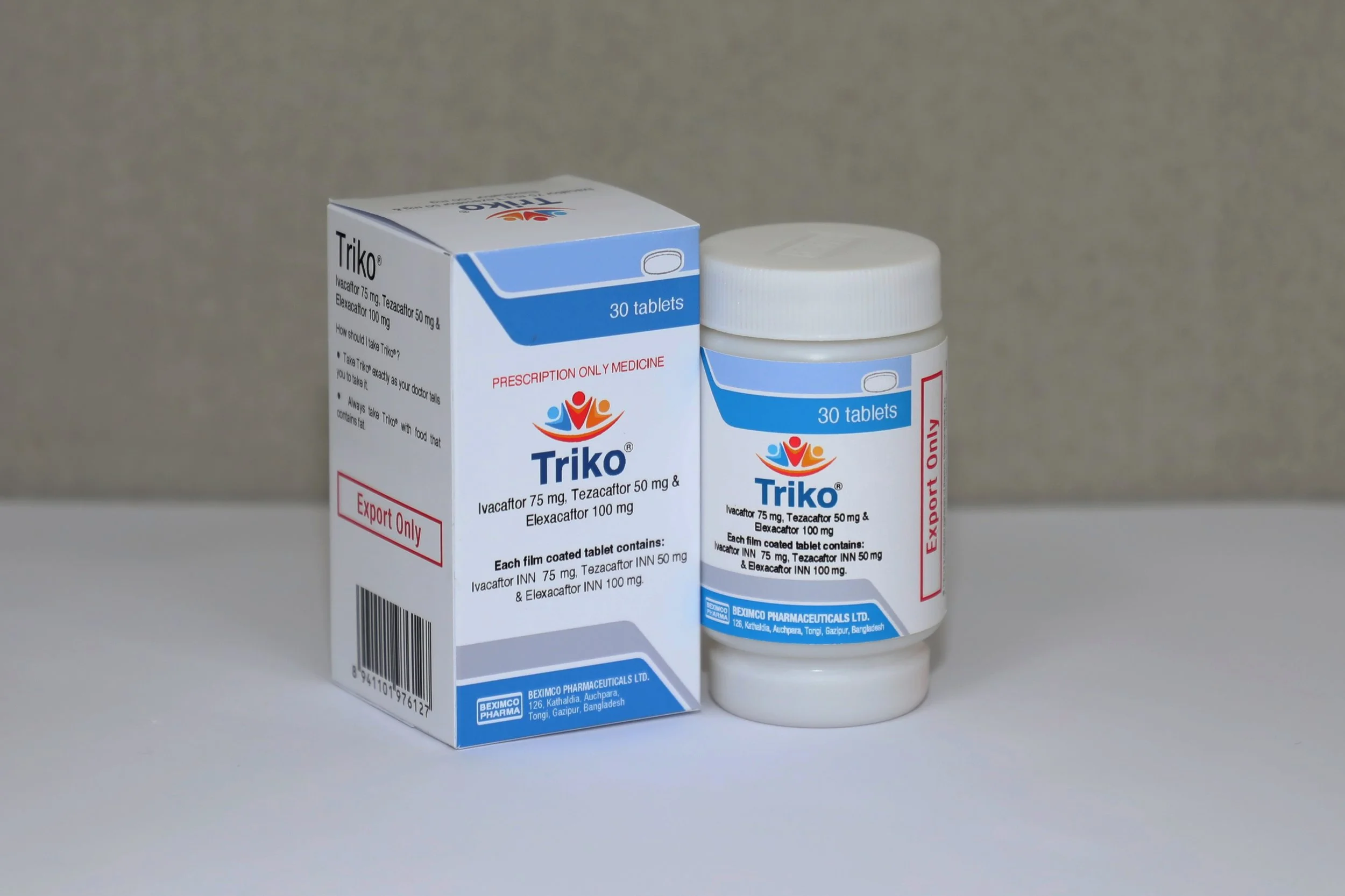
Triko (containing elexacaftor 100mg, tezacaftor 50mg and ivacaftor 150mg) - 30 tablets per pot, costing US$450 per pot, and
Bexdeco (containing ivacaftor 150mg only) - 30 tablets per pot, costing US$150 per pot
Dosage:
An adult patient - or a child aged 6-12 years and over 30kg in weight - will need two pots of Triko (60 tablets in total) and one pot of Bexdeco (30 tablets) every month. Families could also discuss if children in this category could start on a lower dose to check for side-effects and to lower the cost of the treatment, but you would need to discuss this with your CF doctor.
For a child aged 6-12 years and under 30kg in weight, they will need to take half of the adult dose - therefore, two pots of Triko (60 tablets) and one pot of Bexdeco (30 tablets) will last for two months instead of one month.
Dose-sparing dosage (see information below):
For an adult patient - or a child aged 6-12 years and over 30kg in weight, who is also taking a strong CYP3A inhibitor - two Triko tablets are taken on day one and four every week, therefore four Triko tablets are needed per week.
At this dosage, one pot of Triko (30 tablets) will last for 7.5 weeks. Bexdeco is not needed.
Dose-sparing strategy explained:
Clarithromycin is a strong Cytochrome P450 3A (CYP3A) inhibitor that delays liver metabolism of elexacaftor, tezacaftor and ivacaftor (ETI) when administered alongside ETI. The recommended dose adjustment is to reduce ETI to twice weekly when coadministered with a strong CYP3A inhibitor. Left with little choice to save costs, some patients in South Africa opted to exploit this drug interaction on a long-term basis under the supervision of their medical practitioner. You can read the study here.
The study concluded that there was no clinically significant difference in outcomes between the modulator-sparing and standard dosing approaches. The authors of the study propose that modulator-sparing strategies with a strong CYP3A inhibitor may be an acceptable, effective, and cost-saving interim measure to increase access to lifesaving CF modulators in low and middle income countries.
Further questions from the recent webinar:
Would you bring Triko directly to the country without a third party?
We are currently exploring the best way to deliver Triko & Bexdeco to patients that want to buy them. There may be different options - a family member could go to Bangladesh to collect the medicines in person, or it may be possible to have the medicines shipped to your country depending on the specific personal importation rules. We hope to be able to provide clarification on this very soon.
Will there be any problems with customs?
Custom rules will vary from country to country so we recommend that anyone purchasing Triko & Bexdeco should explore their own customs rules and regulations. We will also provide more guidance on this at a later date, once we have finalised the supply logistics for Triko & Bexdeco.
Are there any side effects identified for Triko (similarly as the ones found for Trikafta)?
As Triko & Bexdeco are generic versions of Trikafta & Kalydeco and contain the same active pharmaceutical ingredients (API) we would expect them to have similar side effects.
Is the medicine available to buy at a pharmacy? Do I need a prescription from the doctor to buy it?
CF families and patients who want to buy Triko & Bexdeco will need a prescription. This can be obtained from your CF doctor, who should also be willing to monitor and support the patient whilst they are taking these medicines. However, if it is difficult for a family to obtain a prescription, we may be able to help with this. We will share more details later.
A concern of patients with CF is that they are given a generic drug that does not have the same effect. How to know / understand to address patient fear?
Please go to the CF Buyers Club website here and look at ‘how do we know the generic is safe and effective’.
What date do you have for starting distribution of the medicine?
We are aiming to be able to start distributing the medicines towards the end of 2026. We will let everyone know how and when families can start placing orders.
Besides price, what advantages and disadvantages or differences are there between generic drugs from Bangladesh, Argentina, and perhaps other countries?
If a drug is a generic version of Trikafa there should be very little or no difference between the generic version and the originator product, regardless of where it is produced.
Is there any ongoing process to obtain regulatory prequalification from the PAHO - OPS (Pan American Health Organization)?
Not at the moment but this is something which is being considered.
Does this new medication work for Heterozygotes 508?
Yes, this medicine is suitable for the same genetic mutations as for Trikafta, including patients that are F508del Heterozygotes.
We plan to hold further webinars soon to discuss access strategies, including how to put pressure on governments in specific countries. Please join our mailing list for details of when that meeting will take place, and for further updates about Triko and its distribution